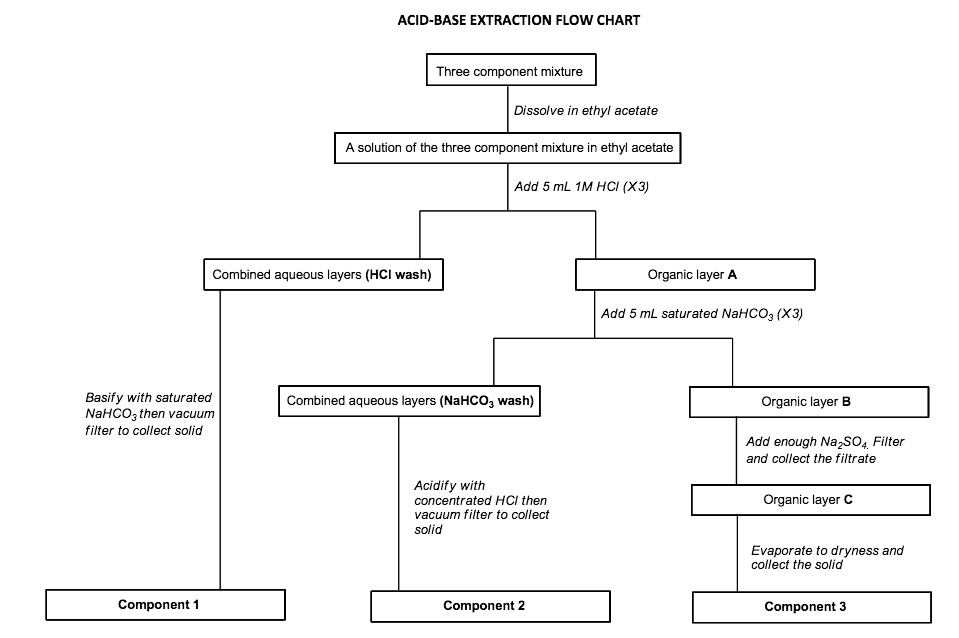
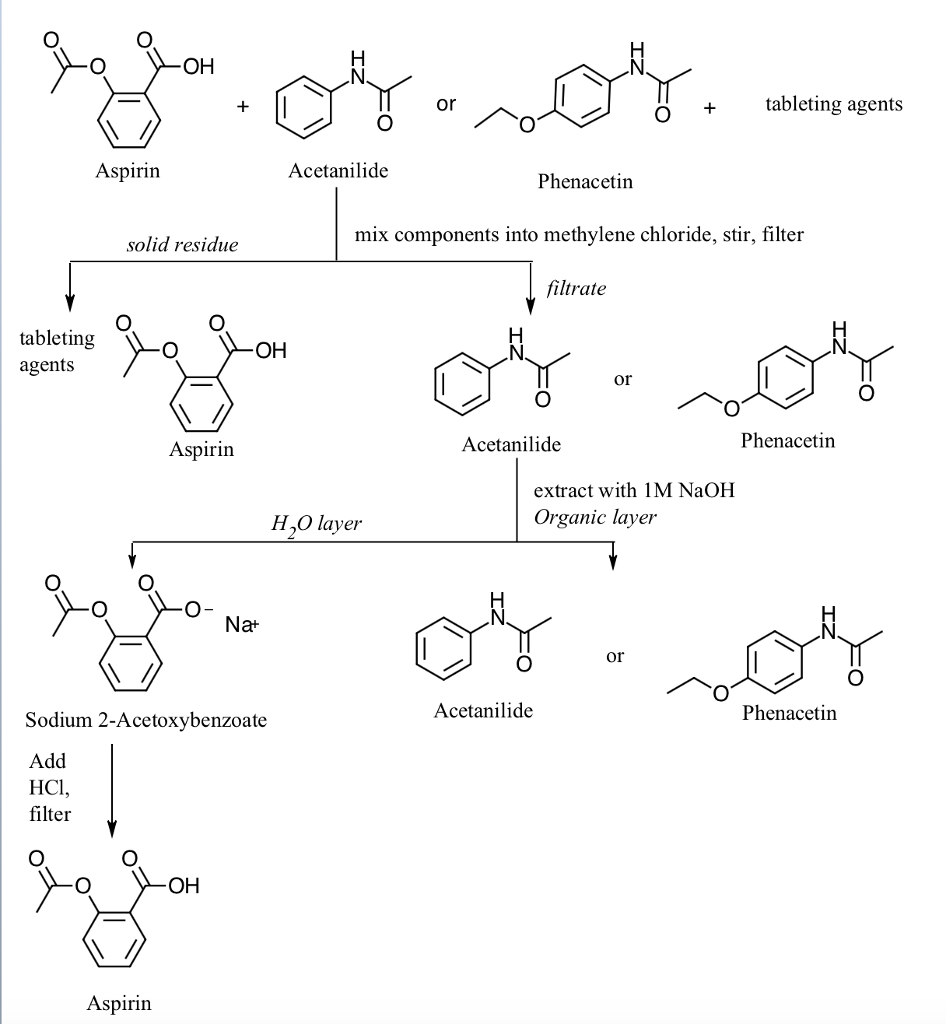
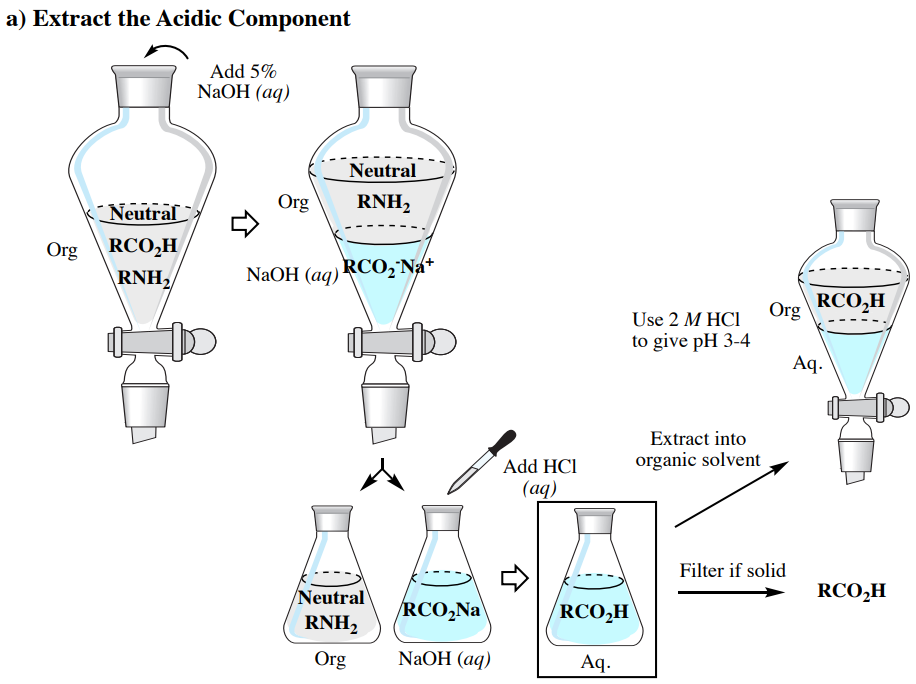
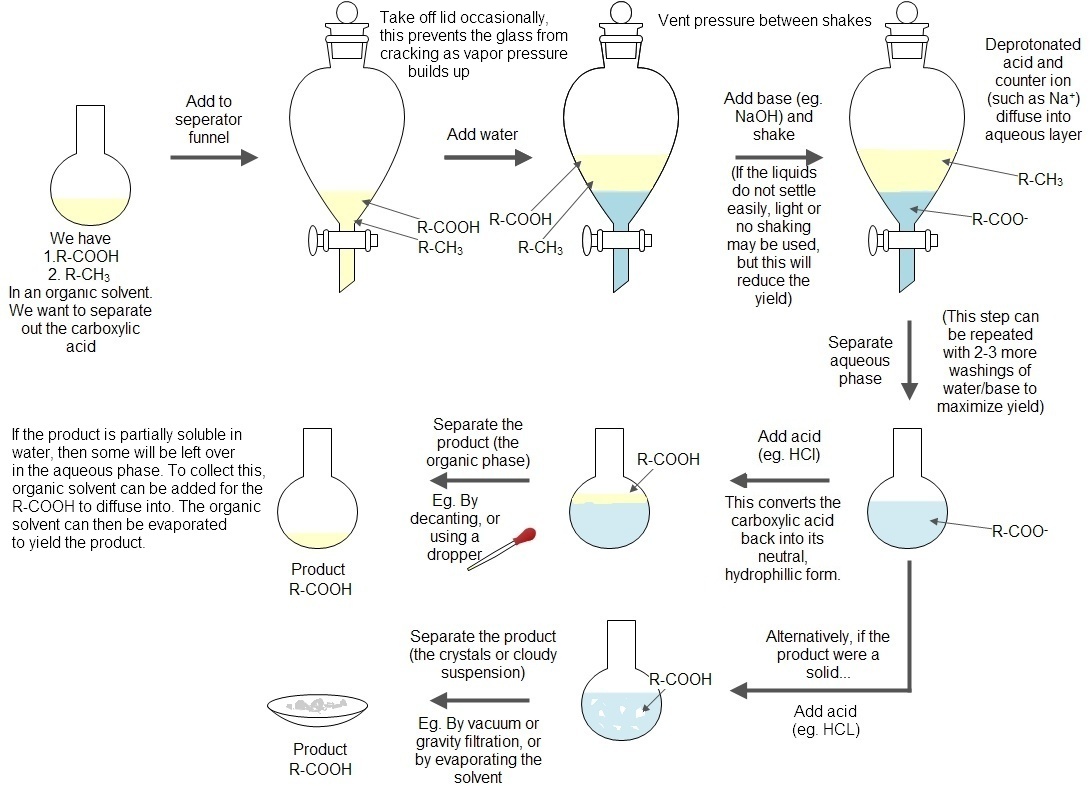
Acid Base Extraction Flow Chart
Acid Base Extraction Flow Chart - Attach your small metal ring to one of the vertical rods on your rack 2. There are three basic types of extraction: Separation of a neutral from a carboxylic acid. Do not ignore this requirement. Phenol will react with a strong base such as sodium hydroxide. We will be performing the first two types of extraction in this experiment. It typically involves different solubility levels in water and an organic solvent. 20 ml of 10% bicarbonate is added to the solution in a separatory funnel where the aqueous Separation of a neutral from a carboxylic acid. Web the flow chart on the next page outlines a general procedure for separating acidic, basic and neutral organic compounds using the principles of the solubility switch. This chart is ideal for use in the lab or in the classroom. The mixture is dissolved in 30 ml of diethyl ether. It typically involves different solubility levels in water and an organic solvent. Separation of a neutral from a carboxylic acid. There are three basic types of extraction: Common ones are ether, ethyl acetate, or dichloromethane. Web chem 344 extraction flowchart. Of acid rco 2 h rco 2 h acid rnh 2 base n neutral extract with 5% naoh organic phase aqueous phase extract with 10% hcl evap. We will be performing the first two types of extraction in this experiment. Attach your small metal ring to one of the vertical rods on your rack 2. Web figure 6 details a full acid/base extraction flow chart that describes the separation of a mixture of an organic acid (ha), organic base (b:) and a neutral organic molecule (n). Do not ignore this requirement. Web two base extraction introduction : This chart is ideal for use in the lab or in the classroom. Attach your small metal ring. Web this flow chart should provide a clear outline of all the steps in the process and should therefore enable you to follow the experiment more easily. Web the flow chart on the next page outlines a general procedure for separating acidic, basic and neutral organic compounds using the principles of the solubility switch. Of acid rco 2 h rco. Common ones are ether, ethyl acetate, or dichloromethane. Of acid rco 2 h rco 2 h acid rnh 2 base n neutral extract with 5% naoh organic phase aqueous phase extract with 10% hcl evap. Aspirin (acetlysalicylic acid) dissolve mixture in about 30ml dichloromethane in erlenmeyer flask and transfer dissolved solution into a separatory funnel. Web via and acid/base reaction. Web we wash the organic reaction mixture with water (acidic, neutral and/or basic) to remove any byproducts or inorganic material. Web via and acid/base reaction the changes in charge and polarity upon reaction for these species can be used to separate them due to their changes in solubility. [study aids] combined acidic extract/combined basic extract [study aids] Aspirin (acetlysalicylic acid). Web chem 344 extraction flowchart. The mixture is dissolved in 30 ml of diethyl ether. Phenol will react with a strong base such as sodium hydroxide. This chart is ideal for use in the lab or in the classroom. 20 ml of 10% bicarbonate is added to the solution in a separatory funnel where the aqueous We will be performing the first two types of extraction in this experiment. In this lab, you'll separate a mixture of cellulose, caffeine, and benzoic acid based on their solubilities in dichloromethane, or dcm, and water. Attach your small metal ring to one of the vertical rods on your rack 2. Web extraction and filtration can separate compounds based on. Common ones are ether, ethyl acetate, or dichloromethane. Web figure 6 details a full acid/base extraction flow chart that describes the separation of a mixture of an organic acid (ha), organic base (b:) and a neutral organic molecule (n). [study aids] combined acidic extract/combined basic extract [study aids] We will be performing the first two types of extraction in this. Web via and acid/base reaction the changes in charge and polarity upon reaction for these species can be used to separate them due to their changes in solubility. The mixture is dissolved in 30 ml of diethyl ether. Web we wash the organic reaction mixture with water (acidic, neutral and/or basic) to remove any byproducts or inorganic material. Web this. The mixture is dissolved in 30 ml of diethyl ether. The acid and base chart is a reference table designed to make determining the strength of acids and bases simpler. This chart is ideal for use in the lab or in the classroom. The general flowchart of the separation is shown below. There are three basic types of extraction: Common ones are ether, ethyl acetate, or dichloromethane. The acid and base chart is a reference table designed to make determining the strength of acids and bases simpler. 20 ml of 10% bicarbonate is added to the solution in a separatory funnel where the aqueous Of acid rco 2 h rco 2 h acid rnh 2 base n neutral extract. Web we wash the organic reaction mixture with water (acidic, neutral and/or basic) to remove any byproducts or inorganic material. Aspirin (acetlysalicylic acid) dissolve mixture in about 30ml dichloromethane in erlenmeyer flask and transfer dissolved solution into a separatory funnel. This chart is ideal for use in the lab or in the classroom. We will be performing the first two types of extraction in this experiment. Web the flow chart on the next page outlines a general procedure for separating acidic, basic and neutral organic compounds using the principles of the solubility switch. Web flow chart of acid base extraction. Common ones are ether, ethyl acetate, or dichloromethane. The acid and base chart is a reference table designed to make determining the strength of acids and bases simpler. Web this flow chart should provide a clear outline of all the steps in the process and should therefore enable you to follow the experiment more easily. Of acid rco 2 h rco 2 h acid rnh 2 base n neutral extract with 5% naoh organic phase aqueous phase extract with 10% hcl evap. Web figure 6 details a full acid/base extraction flow chart that describes the separation of a mixture of an organic acid (ha), organic base (b:) and a neutral organic molecule (n). There are three basic types of extraction: [study aids] combined acidic extract/combined basic extract [study aids] Web via and acid/base reaction the changes in charge and polarity upon reaction for these species can be used to separate them due to their changes in solubility. 20 ml of 10% bicarbonate is added to the solution in a separatory funnel where the aqueous Web chem 344 extraction flowchart.Acid Base Extraction Flow Chart
Acid Base Extraction Flow Chart
ACIDBASE EXTRACTION FLOW CHARTThree component mixtur… SolvedLib
Acid Base Extraction Flow Chart
Solved ACID/BASE EXTRACTION Provide a flow chart detailing
Acid Base Flowchart
Flow Chart Of Acid Base Extraction flow chart
Extraction Flow Chart Organic Chemistry
Visually Tracking AcidBase Extractions Using Colorful Compounds
Acidbase Extraction Flow Chart
Web However, By Taking Advantage Of The Presence Of Acidic And Basic Groups, It Is Sometimes Possible To Achieve Clean Separations Of Mixtures Using A Separating Funnel, An Organic Solvent Such As Ether, And A Sequence Of Extractions With Strong Acids And Bases.
Web Acid/Base Extraction Flow Chart.
In This Lab, You'll Separate A Mixture Of Cellulose, Caffeine, And Benzoic Acid Based On Their Solubilities In Dichloromethane, Or Dcm, And Water.
Separation Of A Neutral From A Carboxylic Acid.
Related Post: