Pressure Temperature Chart Nitrogen
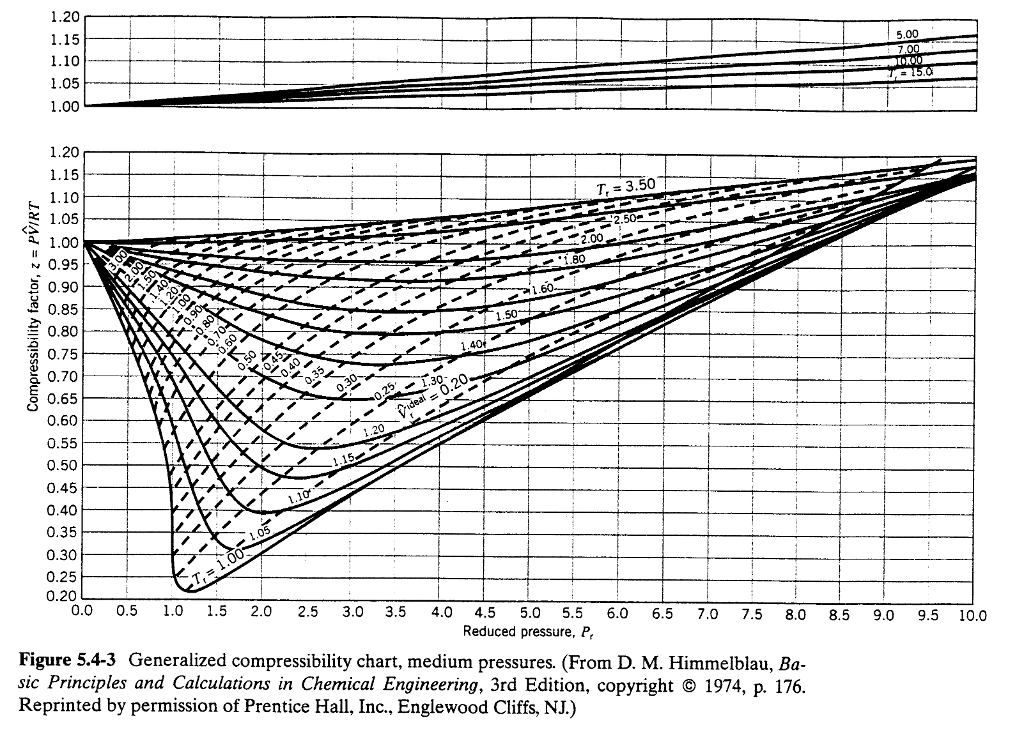
Pressure Temperature Chart Nitrogen - The only way the pressure changed is if there was another gas in the system. This version uses nist refprop for much greater accuracy. Some are constant temperatures, as shown. The model may be used to calculate the thermodynamic properties of mixtures at various compositions including dew and Web the following thermodynamic properties are calculated: Web nitrogen pressure does not change with temp change, you have a leak, even if it changed 1lb you have a leak. Web routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Web most of the values in this table are for data at a constant pressure of 760 torr. Web accurate thermophysical properties are available for several fluids. The approximations are polynomials worked out by the least squares method. Nitrogen n 2 28.013 0.2968 126.2 3.39 0.0899 nitrous oxide n His nitrogen pressure calculator used the ideal gas law to solve for final pressure. The approximations are polynomials worked out by the least squares method. Web nitrogen pressure does not change with temp change, you have a leak, even if it changed 1lb you have a leak. Web for this compound, wtt contains critically evaluated recommendations for: Giauque and clayton, 1933 giauque, w.f.; Web online calculator, figures and tables showing thermal conductivity of nitrogen, n2, at varying temperarure and pressure, si and imperial units. Triple point temperature (crystal 1, liquid, and gas) 30 experimental data points. Pv = nrt, where p, v and t is the pressure, volume and temperature of gas respectively; Web most of the values in this table are for data at a constant pressure of 760 torr. Vapor pressure of solid and liquid. The approximations are polynomials worked out by the least squares method. Pv = nrt, where p, v and t is the pressure, volume and temperature of gas respectively; Web this page relies on the ideal gas law to calculate values of pressure at different temperatures: The fitted expressions have been compared with exper imental. Pv = nrt, where p, v and t is the pressure, volume and temperature of gas respectively; Triple point temperature (crystal 1, liquid, and gas) 30 experimental data points. Web routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. The approximations are polynomials worked out by the least squares method. The only way. The temperature glide runs about 13°f in the evaporator. Web routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Icemeister was curious as to how high pressures would be in his nitrogen cylinder when it was stored in a hot ares. The only way the pressure changed is if there was another gas. Web online calculator, figures and tables showing thermal conductivity of nitrogen, n2, at varying temperarure and pressure, si and imperial units. Web this page relies on the ideal gas law to calculate values of pressure at different temperatures: This version uses nist refprop for much greater accuracy. His nitrogen pressure calculator used the ideal gas law to solve for final. This version uses nist refprop for much greater accuracy. Web the reduced density and reduced temperature. Pv = nrt, where p, v and t is the pressure, volume and temperature of gas respectively; Web routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Giauque and clayton, 1933 giauque, w.f.; The temperature glide runs about 13°f in the evaporator. Clayton, j.o., heat capacity and entropy of nitrogen. These data include the following: Web the reduced density and reduced temperature. This version uses nist refprop for much greater accuracy. Web this page relies on the ideal gas law to calculate values of pressure at different temperatures: Vapor pressure of solid and liquid. Pv = nrt, where p, v and t is the pressure, volume and temperature of gas respectively; Web online calculator, figures and tables showing thermal conductivity of nitrogen, n2, at varying temperarure and pressure, si and imperial. Web for this compound, wtt contains critically evaluated recommendations for: Otto, j., vapor pressure curves and triple points in the temperature region from 14 to 90 k, phys. Icemeister was curious as to how high pressures would be in his nitrogen cylinder when it was stored in a hot ares. Triple point temperature (crystal 1, liquid, and gas) 30 experimental. Triple point temperature (crystal 2, crystal 1. Some are constant temperatures, as shown. Clayton, j.o., heat capacity and entropy of nitrogen. Web routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Web the reduced density and reduced temperature. Web nitrogen pressure does not change with temp change, you have a leak, even if it changed 1lb you have a leak. The temperature glide runs about 13°f in the evaporator. Molar mass, constant, temperature, pressure, volume, substance formula mkg/kmol rkj/kg·k* k mpa m3/kmol. As discussed in chapter 9, there should not be much difference between the constants obtained either. Web for this compound, wtt contains critically evaluated recommendations for: Web routines for the physical properties of nitrogen liquid and gas (1 to 6 bar) are presented. Vapor pressure of solid and liquid. The only way the pressure changed is if there was another gas in the system. Pv = nrt, where p, v and t is the pressure, volume and temperature of gas respectively; Giauque and clayton, 1933 giauque, w.f.; Web online calculator, figures and tables showing thermal conductivity of nitrogen, n2, at varying temperarure and pressure, si and imperial units. Web nitrogen and methane viscosities are directly proportional to the pressure with a correlation coefficient of 4% and 97% respectively, and inversely proportional to temperature with a correlation. The fitted expressions have been compared with exper imental data, and the accuracy is. Nitrogen n 2 28.013 0.2968 126.2 3.39 0.0899 nitrous oxide n The approximations are polynomials worked out by the least squares method. Molar mass, constant, temperature, pressure, volume, substance formula mkg/kmol rkj/kg·k* k mpa m3/kmol. This version uses nist refprop for much greater accuracy. Web this page relies on the ideal gas law to calculate values of pressure at different temperatures: Web nitrogen pressure does not change with temp change, you have a leak, even if it changed 1lb you have a leak. Web accurate thermophysical properties are available for several fluids.Nitrogen Pressure Chart A Visual Reference of Charts Chart Master
Nitrogen Pressure Chart A Visual Reference of Charts Chart Master
Nitrogen Phase Diagram Pressure Temperature General Wiring Diagram
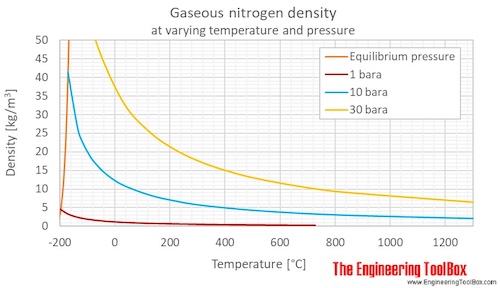
Nitrogen Density and Specific Weight vs. Temperature and Pressure
Nitrogen Density and Specific Weight vs. Temperature and Pressure
Nitrogen Pressure Temperature Chart
Nitrogen Density and Specific Weight vs. Temperature and Pressure
Nitrogen Density and Specific Weight vs. Temperature and Pressure
Critical temperaturepressure curve for nitrogen transmutation and
Phase diagrams for nitrogen. At high temperature and pressure, a plasma
Some Are Constant Temperatures, As Shown.
Icemeister Was Curious As To How High Pressures Would Be In His Nitrogen Cylinder When It Was Stored In A Hot Ares.
Triple Point Temperature (Crystal 2, Crystal 1.
Web The Reduced Density And Reduced Temperature.
Related Post: