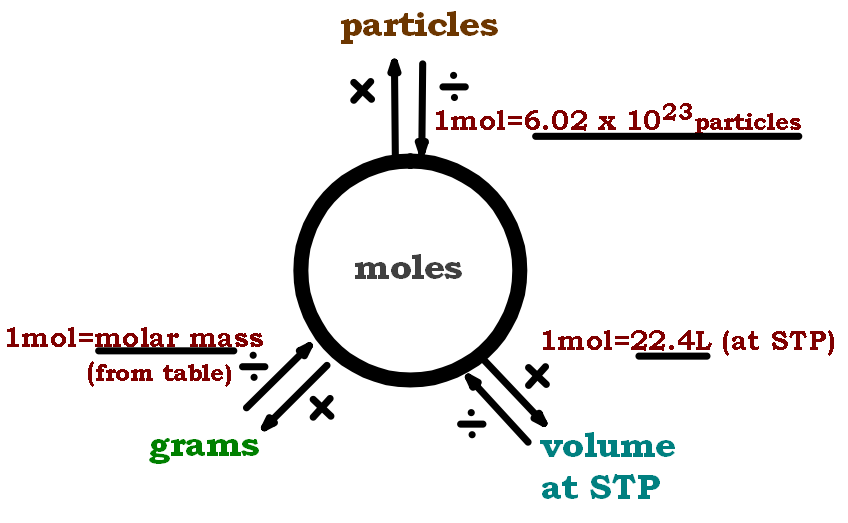
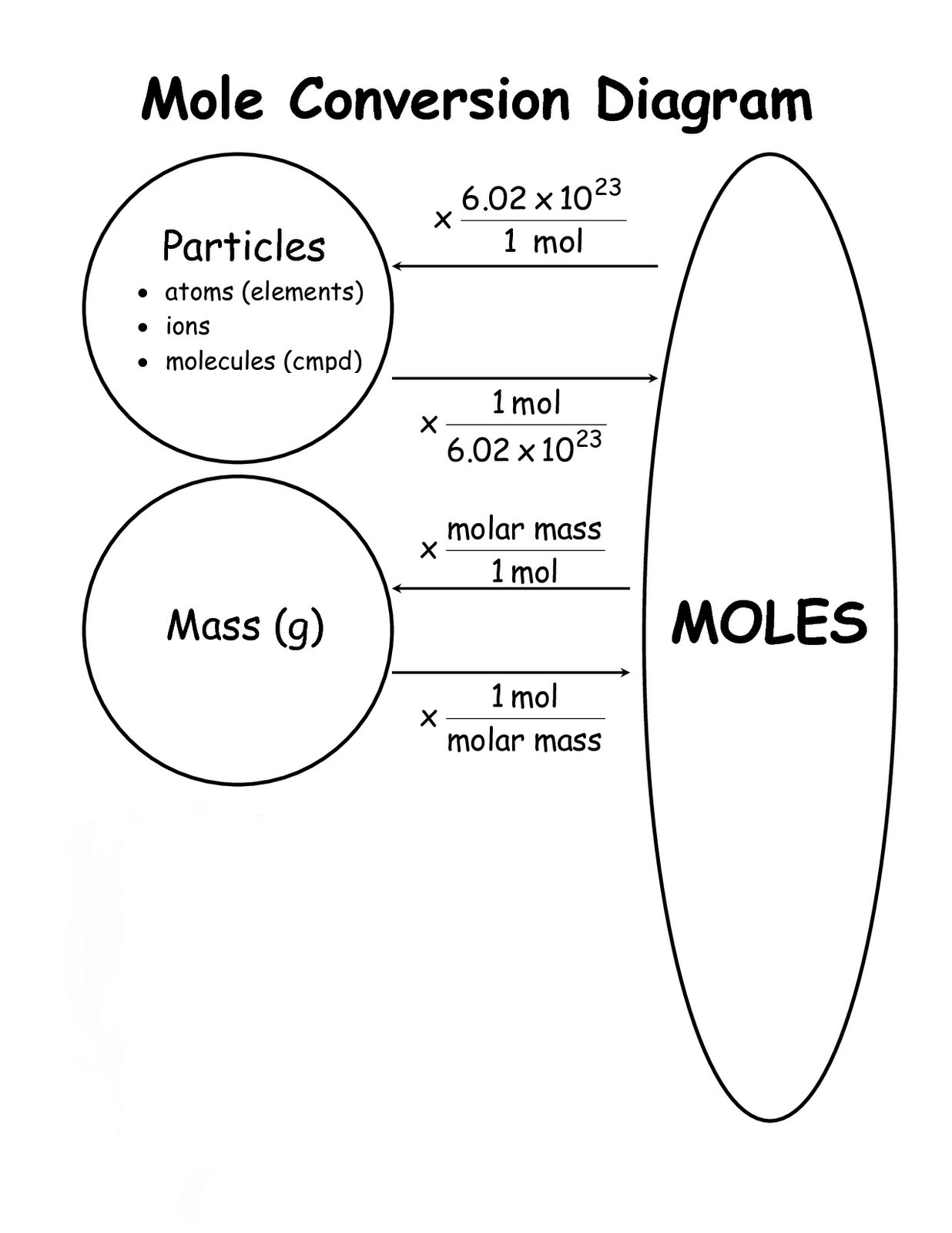
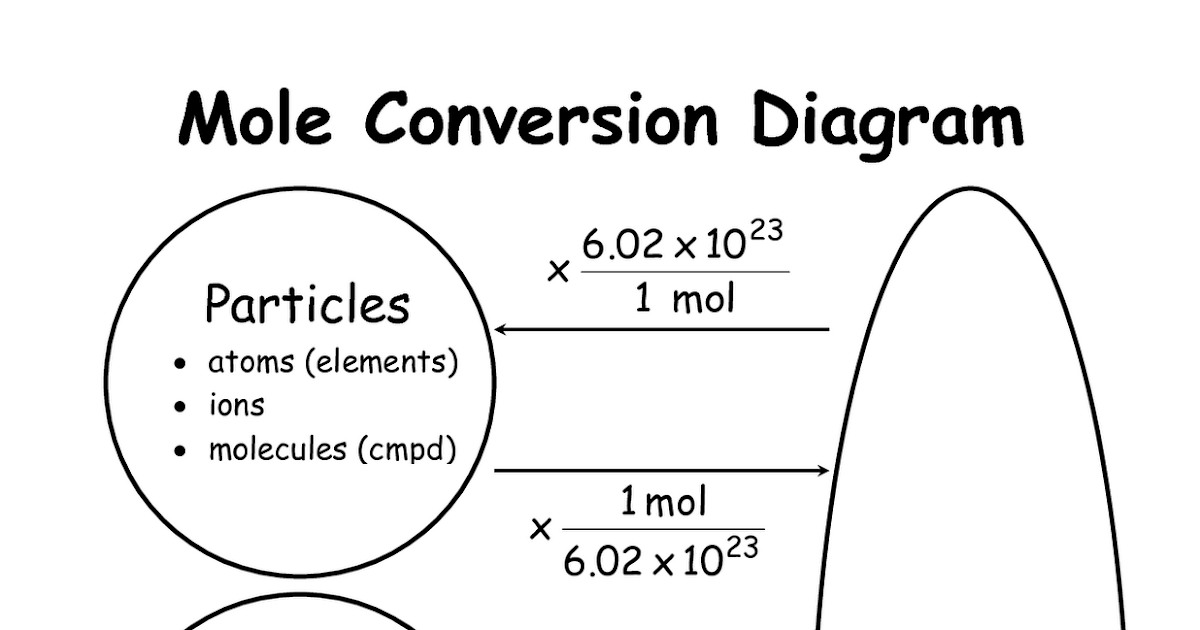
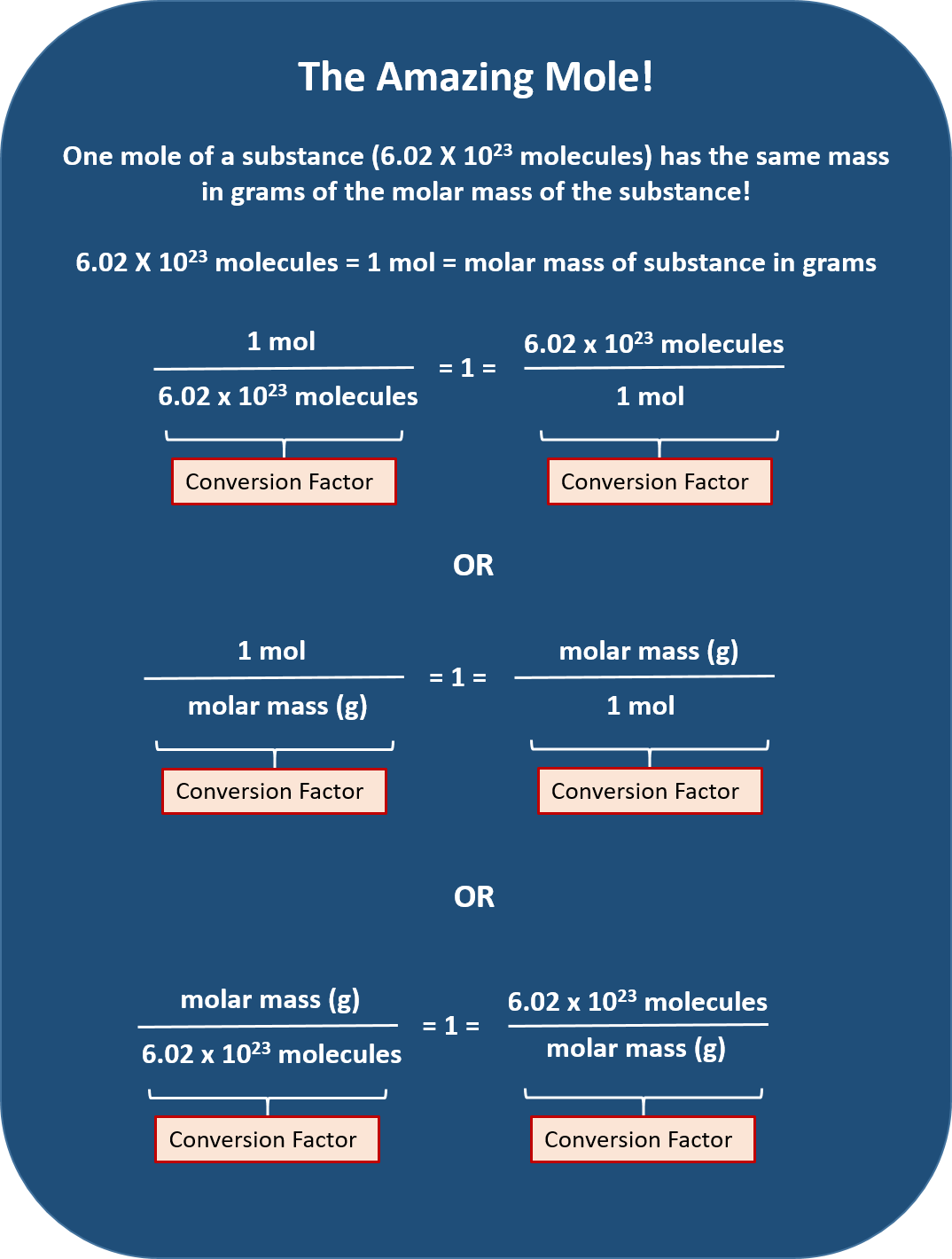
Mol Conversion Chart
Mol Conversion Chart - The molecular weight of sodium chloride, nacl , is 58.44 g mol. A mole is an avogadro's number (6.023 × 10 23) of elementary entities (eg, atoms, ions, molecules); Convert between moles and grams. Web perform conversions between mass and moles of a substance. Why does the calculator use 56.6% weight percentage instead of 28% for ammonium. Web you can calculate the molar mass of a molecule by summing the molar masses of the composing atoms with the help of the periodic table. Web figure \(\pageindex{1}\) is a chart for determining what conversion factor is needed, and figure \(\pageindex{2}\) is a flow diagram for the steps needed to perform. How many moles of salt are in 13.8 g of sodium chloride? Web all you need to do is correctly enter your formula, choose whether you want a conversion from grams to moles or a conversion from moles to grams, and, in case of g to mol,. Web moles are a type of unit conversion used in chemistry to measure the amount of substances. A mole is an avogadro's number (6.023 × 10 23) of elementary entities (eg, atoms, ions, molecules); Multiply your initial mole value by the molar mass of the compound as determined by the periodic table. Formula weight (daltons) moles = grams from. Web our moles to grams converter makes it easy to convert between molecular weight, mass, and moles. The mole map is a powerful tool to visualize mole conversions. Convert grams to moles effortlessly with this tool. Web this mole calculator helps to determine the number of moles, mass, or molecular weight of a substance. The si unit of concentration (molar) is mole / cubic meter [ mol / m3 ] total number of units: Web you can calculate the molar mass of a molecule by summing the molar masses of the composing atoms with the help of the periodic table. How many moles of salt are in 13.8 g of sodium chloride? The molecular weight of sodium chloride, nacl , is 58.44 g mol. The mass of 1 mole of a substance is. Formula weight (daltons) moles = grams from. Web our moles to grams converter makes it easy to convert between molecular weight, mass, and moles. Web figure \(\pageindex{1}\) is a chart for determining what conversion factor is needed, and figure. A mole is an avogadro's number (6.023 × 10 23) of elementary entities (eg, atoms, ions, molecules); The molecular weight of sodium chloride, nacl , is 58.44 g mol. Chemistry just became that little bit easier! Formula weight (daltons) moles = grams from. Web moles are a type of unit conversion used in chemistry to measure the amount of substances. Web all you need to do is correctly enter your formula, choose whether you want a conversion from grams to moles or a conversion from moles to grams, and, in case of g to mol,. Web figure \(\pageindex{1}\) is a chart for determining what conversion factor is needed, and figure \(\pageindex{2}\) is a flow diagram for the steps needed to. Web perform conversions between mass and moles of a substance. Use this grams to moles calculator. Web you can calculate the molar mass of a molecule by summing the molar masses of the composing atoms with the help of the periodic table. Web converting from moles to mass (grams): Convert grams to moles effortlessly with this tool. Web whether you’re converting from moles to grams, moles to volume, or moles to particles (atoms or molecules), use this quick guide to remind you of how to do each type of mole. Web this free moles to grams calculator can instantly convert moles to grams and determine how many atoms/molecules are present in these grams. Web converting from moles. Web moles, milligrams, and milliequivalents: Web this free moles to grams calculator can instantly convert moles to grams and determine how many atoms/molecules are present in these grams. Web moles are a type of unit conversion used in chemistry to measure the amount of substances. Convert from mass or moles of one substance to mass or moles of another substance. Previously, you learned to balance chemical equations by comparing the numbers of. One mole is equal to 6.022 x 10^23 atoms, molecules or other particles. Web this mole calculator helps to determine the number of moles, mass, or molecular weight of a substance. Web figure \(\pageindex{1}\) is a chart for determining what conversion factor is needed, and figure \(\pageindex{2}\) is. Convert from mass or moles of one substance to mass or moles of another substance in a. The molecular weight of sodium chloride, nacl , is 58.44 g mol. Convert from mass or moles of one substance to mass or moles of another substance in a chemical reaction. One mole is equal to 6.022 x 10^23 atoms, molecules or other. Formula weight (daltons) moles = grams from. Web you can calculate the molar mass of a molecule by summing the molar masses of the composing atoms with the help of the periodic table. If you are struggling, it will quickly help you understand how to convert from moles, to grams,. Web perform conversions between mass and moles of a substance.. Web you can calculate the molar mass of a molecule by summing the molar masses of the composing atoms with the help of the periodic table. Web mole conversion chart national mole day october 23. The mole map is a powerful tool to visualize mole conversions. Multiply your initial mole value by the molar mass of the compound as determined. Convert from mass or moles of one substance to mass or moles of another substance in a. Web figure \(\pageindex{1}\) is a chart for determining what conversion factor is needed, and figure \(\pageindex{2}\) is a flow diagram for the steps needed to perform. Follow along, and we’ll show you how to convert mass to. The mass of 1 mole of a substance is. Web converting from moles to mass (grams): Web you might be wondering how you can calculate the number of moles given a mass, volume, or amount of substance. Web all you need to do is correctly enter your formula, choose whether you want a conversion from grams to moles or a conversion from moles to grams, and, in case of g to mol,. Web this free moles to grams calculator can instantly convert moles to grams and determine how many atoms/molecules are present in these grams. Web perform conversions between mass and moles of a substance. If you are struggling, it will quickly help you understand how to convert from moles, to grams,. Μg mg g dag kg t oz lb. Convert between moles and grams. Web you can calculate the molar mass of a molecule by summing the molar masses of the composing atoms with the help of the periodic table. Convert grams to moles effortlessly with this tool. How many moles of salt are in 13.8 g of sodium chloride? The si unit of concentration (molar) is mole / cubic meter [ mol / m3 ] total number of units:Unit 7 Math of Chemistry Ivy Way Science
Chemistry Mysteries Mole Conversions
Chemistry Mysteries Mole Conversions
Molarity, Molality, Mass Fraction Conversion Formula Math Encounters Blog
Mole and mass conversions Science, Chemistry, Stoichiometry ShowMe
1Mole Flow Chart.cwk (WP)
Chapter 6 Quantities in Chemical Reactions Chemistry
PPT Chemistry 20 Mole Conversions PowerPoint Presentation, free
Chemistry Cheat Sheet Mole Conversion Chart Download Printable PDF
Molar Mass Conversion Chart
The Molecular Weight Of Sodium Chloride, Nacl , Is 58.44 G Mol.
Use This Grams To Moles Calculator.
Web This Mole Calculator Helps To Determine The Number Of Moles, Mass, Or Molecular Weight Of A Substance.
Web Our Moles To Grams Converter Makes It Easy To Convert Between Molecular Weight, Mass, And Moles.
Related Post: